Molarity Of 5 Acetic Acid
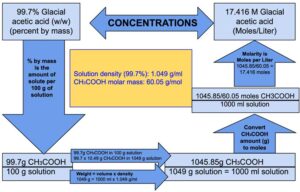
Glacial acetic acid is a clear colorless liquid. A 99.7% (west/w) concentrated glacial acetic acid can be obtained from unlike suppliers. A 99.7% (w/w) glacial acetic acrid ways that 100 g of glacial acetic acrid contains 99.vii thou of acerb acrid. The density of glacial acetic acrid is 1.049 grand/ml at 25°C which means that the weight of the i ml of glacial acetic acid is 1.049 grams at 25°C. Molarity refers to the number of moles of the solute present in 1 liter of solution.
Adding procedure:
| Known values | |
| Density of glacial acetic acid | ane.049 grand/ml |
| Molar mass of acerb acid (CH3COOH) | 60.05 g/mole |
| Concentration of glacial acerb acid | 99.7% (% by mass, wt/wt) |
Stride ane: Since molarity refers to number of moles in 1000 ml solution, calculate the weight of 1000 ml Glacial acetic acid using the following formula:
Formula:
or
weight = Density ten book
Weight = thou x 1.049 = 1049
Weight of m ml Glacial acetic acid is 1049 g.
Pace 2: Calculate how much CH three COOH is in 1049 thou of Glacial acerb acid.
100 g Glacial acetic acid contains: 99.7 yard of CH3COOH
1 thousand Glacial acetic acid will contain: 99.7/100 g of CH3COOH
1049 g Glacial acetic acid volition contain: 1049 x 99.7/ 100 = 1045.85 g of CH3COOH
Note: Since the volume of 1049 one thousand of Glacial acetic acid is thou ml, nosotros can say that 1045.85 m of CH3COOH is nowadays in 1000 ml of Glacial acetic acid.
Pace iii: Calculate the number of moles of acerb acrid nowadays in 1045.85 grams of acetic acrid.
60.05 grams of acetic acid = i mole
This ways 17.416 moles of CH3COOH is present in m ml of Glacial acerb acid.
Therefore, nosotros can say that one liter of glacial acerb acid contains 17.416 moles or in other words molarity of glacial acerb acrid (99.vii% westward/westward, density i.049 k/ml) is equal to 17.416.
Use Figurer to calculate the Molarity of full-bodied acetic acid (CH3COOH) when concentration is given in % by mass (w/west)
The molecular weight of Acetic acid (CH3COOH): 60.05 g/mol
Concentration of acetic acid : % (wt/wt) (Change the % (wt/wt) concentration) Density of glacial acerb acrid: thousand/ml (Change the density) Molarity of acetic acrid: 17.416 K Was this post helpful?
Molarity Of 5 Acetic Acid,
Source: https://www.laboratorynotes.com/how-to-calculate-molarity-of-glacial-acetic-acid-99-7-w-w/
Posted by: hawkinsousioner.blogspot.com


0 Response to "Molarity Of 5 Acetic Acid"
Post a Comment